 Horizon Therapeutics currently carries a Zacks Rank #3 (Hold). Horizon Therapeutics plc price-consensus-eps-surprise-chart | Horizon Therapeutics plc Quote Horizon Therapeutics plc Price, Consensus and EPS Surprise The company expects to begin a pivotal phase IIb study on the same in the first half of 2021. and its development-stage oral selective lysophosphatidic acid 1 receptor (LPAR1) antagonist, CZN001 (renamed HZN-825), for the treatment of diffuse cutaneous systemic sclerosis (dcSSc). In April, Horizon Therapeutics acquired the privately-held, biopharma company Curzion Pharmaceuticals, Inc. Last month, the company announced that Procysbi, currently available in the United States, can be ordered in 75 mg and 300 mg dosage strengths for adults and children aged one year and above, suffering nephropathic cystinosis. This new dosage provides another administration option for patients in addition to the currently available Procysbi capsules. In February, the FDA approved Procysbi delayed-release oral granules in packets for adults and children aged one year and above, living with nephropathic cystinosis. Tepezza is the first and the only FDA-approved medicine for the treatment of TED, a serious, progressive and vision-threatening rare autoimmune disease. Teprotumumab will be marketed under the brand name Tepezza. In January 2020, the FDA approved teprotumumab-trbw for the treatment of thyroid eye disease (TED), well ahead of the set action date of Mar 8, 2020. Tepezza’s full-year net sales are estimated to be more than $200 million compared to its previous outlook of $30-$40 million. The company now expects 2020 net sales between $1.40billion and $1.45 billion, which were earlier projected in the range of $1.40-$1.42 billion. The updated view assumes that healthcare activity will be resumed in the second half of 2020. Per the company, owing to the strong uptake of Tepezza and its sunstantially higher revenues that more than offset the expected adversity from the COVID-19 pandemic, Horizon Therapeutics raised its full-year net sales guidance. 
Per the company, this strong start for Tepezza was owing to significantly higher number of patients beginning therapy than initially expected.įirst-quarter 2020 net sales of the Inflammation segment were $110.5 million, down 3% year over year.Īdjusted R&D and SG&A expenses were 5.8% and 69.6% of net sales, respectively, during the quarter. 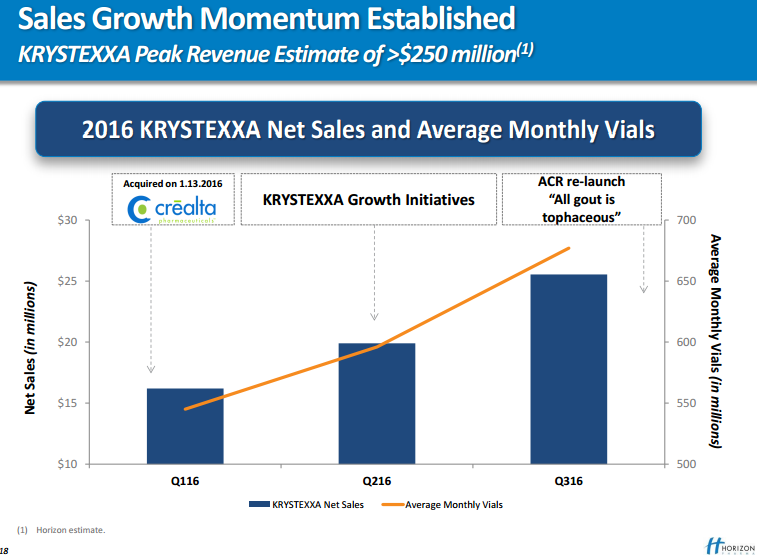
Tepezza generated net sales worth $23.5 million in the first quarter, exceeding management’s expectations. Krystexxa sales soared 78% year over year to $93.3 million. 
Sales of the orphan segment, the company’s strategic growth unit, were $245.4 million, up 47% from the prior-year figure, driven by continued growth of Krystexxa, Ravicti and the launch of Tepezza. Notably, from the first quarter of 2020 onward, the orphan and rheumatology segment was renamed the orphan segment. It reports financial results under two separate segments, namely orphan and rheumatology, and Inflammation (previously known as the primary care segment). The company realigned its structure to operate two separate businesses, effective second-quarter 2018. 
Horizon Therapeutics’ share price has rallied 17.7% year to date compared with the industry’s increase of 3.7%. Last May, the company’s shareholders approved the change in its name to Horizon Therapeutics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
